Benefits
Technology
Specifications
Tour
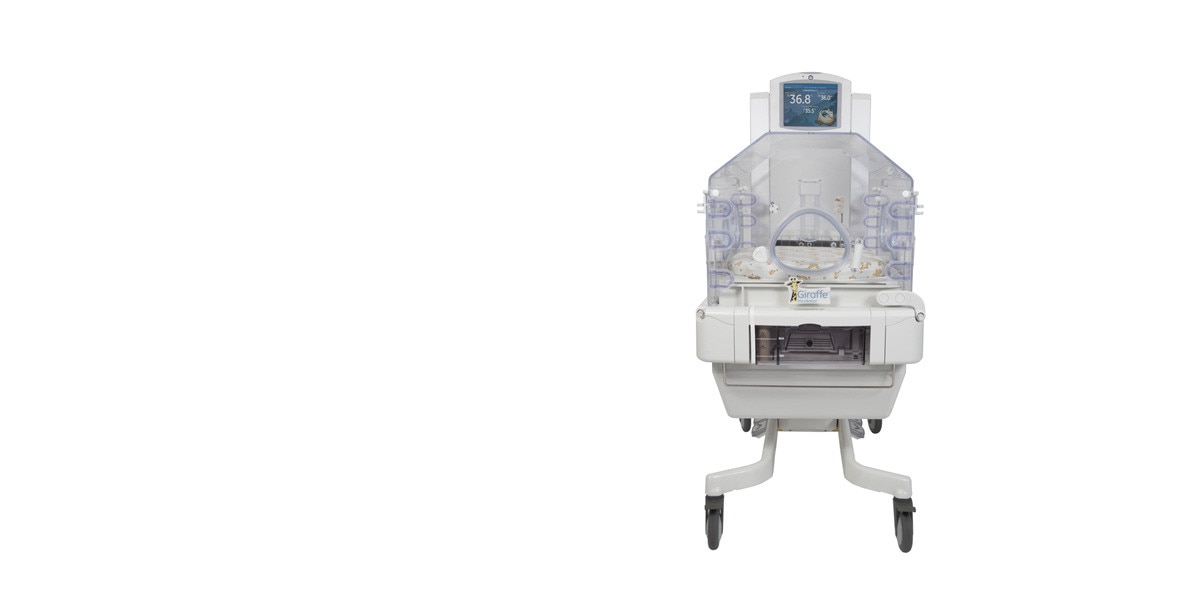
Family-Centered Care
Elevating base designed to accommodate standing caregivers and seated family. Pass-through drawer allows full-seated access to promote crucial parent-baby bonding. Side door panels can be removed completely for greater access. Parent-friendly away screen display.
Thermoregulation
Bi-Directional Airflow through double walls provides a stable thermal environment. Servo-controlled Humidity System designed with easy cleaning in mind to protect against infection. Pre-heat mode at 25% power, with silenced alarms, for admission preparation.
Comfort Zone
Large color display (10.4”) provides guidance for initial settings incubator air temperature for individual patients (derived from Sauer, Dane & Visser research).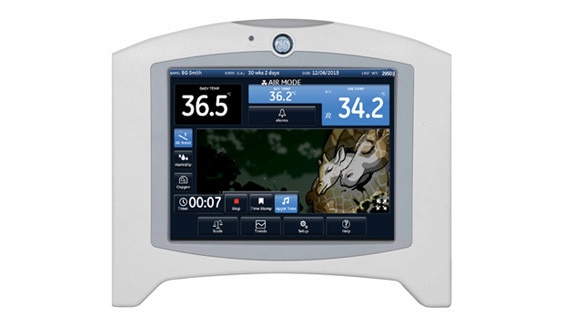
Patient Access
Dual access door panels and portholes allow access even when the bed is tilted 12º. Door panels can be removed for easier access to patients and cleaning. Baby Susan mattress with full 360° rotation.
Air Boost
Helps protect the infant from heat loss when door panels are opened.
360 View
Related
Gallery
Giraffe Incubator Carestation
Giraffe Incubator Carestation
Education & Training
Bangalore 560067,
Karnataka, India
CIN: U33111KA1990PTC01606
