Indications
Reimbursement
See Important Safety Information about AdreView at the bottom of this page.
Please also read the Full Prescribing Information.
GE Healthcare Medical Diagnostics Reimbursement
GE Healthcare advocates for timely patient access to medically appropriate, high quality imaging services, provided by knowledgeable and skilled providers of care. We recognize the significant impact that reimbursement policies can have on you and your practice and strive to provide accurate and reliable information to help you navigate the reimbursement process.
Reimbursement Information for Radiopharmaceuticals: Medicare Coding and Payment for Radiopharmaceuticals Used in Nuclear Medicine Procedures
Coding and Payment Information
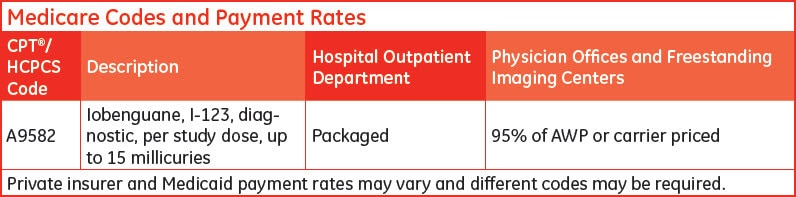
Under HOPPS, Medicare will continue "packaging" for diagnostic radiopharmaceuticals into the payment for the associated procedure.
Diagnostic radiopharmaceuticals are separately payable in physician offices and freestanding imaging centers. Payment is based on 95% of the average wholesale price (AWP) or established by local Medicare contractors (carrier priced).
Physicians performing nuclear medicine procedures using diagnostic radiopharmaceuticals should report the appropriate HCPCS code for the product along with the appropriate CPT® code(s) for the procedure(s). Physicians should make sure the number of units reported is consistent with the quantity of a radiopharmaceutical given to complete the nuclear medicine imaging study.
Professional Resources and Disclosures
Indications and Important Safety Information
JB01343IN
Bangalore 560067,
Karnataka, India
CIN: U33111KA1990PTC01606
